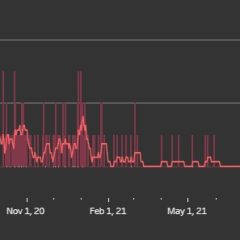
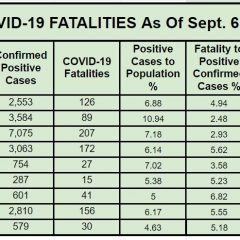
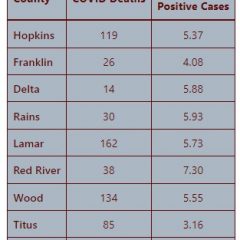
13 Hopkins County COVID Deaths So Far In 2022, 166 Since March 2020
While many states and countries are lowering and, in some cases, even eliminating COVID-19 restrictions altogether, the virus has surged in other areas. After almost 2 full years, many have become weary of the extra precautions, whether required vaccinations, frequent testing, booster shots, masking, lockdowns, restricted gatherings or frequent sanitizing and extra cleansing with virus-killing substances. Health care and educational...
FDA Approves Second COVID-19 Vaccine
News Release – Monday, Jan. 31, 2022 Moderna To Be Marketed as Spikevax COVID-19 Vaccine For Ages 18+ Today (Jan. 31, 2022), the U.S. Food and Drug Administration approved a second COVID-19 vaccine. The vaccine has been known as the Moderna COVID-19 Vaccine; the approved vaccine will be marketed as Spikevax for the prevention of COVID-19 in individuals 18 years of age and older. Key points Spikevax meets the FDA’s rigorous...
FDA Acts To Expand Use Of Pfizer COVID-19 Vaccine
FDA Jan. 3, 2022 News Release Today, the U.S. Food and Drug Administration amended the emergency use authorization (EUA) for the Pfizer-BioNTech COVID-19 Vaccine to: Expand the use of a single booster dose to include use in individuals 12 through 15 years of age.Shorten the time between the completion of primary vaccination of the Pfizer-BioNTech COVID-19 Vaccine and a booster dose to at least five months.Allow for a third primary...
FDA Expands Eligibility For COVID-19 Vaccine Booster For All Individuals 18 Or Older
FDA News Release – November 19, 2021 Today, the U.S. Food and Drug Administration amended the emergency use authorizations (EUA) for both the Moderna and Pfizer-BioNTech COVID-19 vaccines, authorizing use of a single booster dose for all individuals 18 years of age and older after completion of primary vaccination with any FDA-authorized or approved COVID-19 vaccine. The Centers for Disease Control and Prevention’s (CDC)...
FDA Authorizes Pfizer-BioNTech COVID-19 Vaccine For Emergency Use In Children 5 Through 11 Years of Age
Friday, Oct. 29, 2021 USFDA New Release Today, the U.S. Food and Drug Administration authorized the emergency use of the Pfizer-BioNTech COVID-19 Vaccine for the prevention of COVID-19 to include children 5 through 11 years of age. The authorization was based on the FDA’s thorough and transparent evaluation of the data that included input from independent advisory committee experts who overwhelmingly voted in favor of making the...
FDA Takes Additional Actions On Use Of A Booster Dose For COVID-19 Vaccines
US FDA News Release The U.S. Food and Drug Administration Wednesday, Oct. 20, took action to expand the use of a booster dose for COVID-19 vaccines in eligible populations. The agency is amending the emergency use authorizations (EUA) for COVID-19 vaccines to allow for the use of a single booster dose as follows: The use of a single booster dose of the Moderna COVID-19 Vaccine that may be administered at least 6 months after...
Product Recalls: Infant Rice Cereal, Lidocaine, Dog Food, Trail Mix
Time to check the fridge and cabinets to see if you have any of the products recalled over the last week due to possible cross contamination, undeclared potential allergens or high levels of some ingredients including infant rice cereal, lidocaine, dog food and trail mix. Baby Rice Cereal While no illnesses related to the product lots had been reported as of Oct. 8, 2021, Maple Island Inc. opted to issue a voluntary recall of three...
DSHS Announces 126th Hopkins County COVID Death
186 New Cases, 118 Recoveries, 292 Active Cases Of Coronavirus Reported So Far In September Texas Department of State Health Services had announced the 126th Hopkins County COVID death Monday. A total of 186 new cases, 118 additional recoveries and one coronavirus death have been reported for Hopkins County during the first 6 days of September 2021, leaving 392 Hopkins County residents who still actively had the virus at noon on Labor...
Pfizer COVID-19 Vaccine Approved By FDA
The U.S. Food and Drug Administration on Monday fully approved the first COVID-19 vaccine. The vaccine has been known as the Pfizer-BioNTech COVID-19 Vaccine, and will now be marketed as Comirnaty (koe-mir’-na-tee), for the prevention of COVID-19 disease in individuals 16 years of age and older. Pfizer-BioNTech COVID-19 Vaccine receives full FDA approval The vaccine will also continue to be available under emergency use...
3 COVID-19 Deaths, 25 New Cases & 27 Recoveries Reported Aug. 17, 2021, For Hopkins County
The number of COVID-19 cases continues to swell in Hopkins County, with 25 new COVID-19 cases reported Aug. 17, three additional deaths and 27 recoveries reported on Tuesday, 2021. That leaves 244 active COVID-19 cases among Hopkins County residents on Aug. 17. Also across the region, only 1 ICU hospital bed was available on Monday. COVID-19 Deaths Across the state, only 96 new COVID-19 fatalities were recorded by Texas Department of...